|
|
Iminosugars Section
Contribution to iminosugars field:
What is an iminosugar?
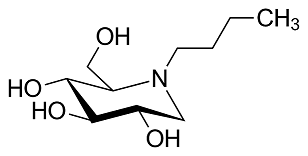
Iminosugars are low molecular weight sugar analogs, where an oxygen atom has been replaced by nitrogen atom in the ring.
This apparently simple substitution give rise many synthetic challenges and creates more opportunities for more significant biological activities.

In May 1991 I was an undergraduate student of prof. Nicotra at Milan University, Italy.
I was working on my degree thesis in organic chemistry and I was stuck in the synthesis of the molecule 9,
when I get the molecule 10 thanks to serendipity.
I remember that when I saw the TLC (thin layer chromatography) of molecule 10, it showed a good yeald, so I sent a sample to NMR analysis.
When I saw the NMR and quantitative elemental analysis I drew the pyrrolidine cycle on the NMR spectra, a molecule that I have never seen.
I showed the NMR spectra to Nicotra and he said happily: "I have already seen it. I offer you a coffee".
For this research, Nicotra offered me an espresso coffee at Wanda's bar in via Celoria/Golgi in Milan.
thesis_page23 (1991)
thesis_page24 (1991)
thesis_page50 (1991)
thesis_page56 (1991)
thesis_page57 (1991)
thesis_page58 (1991)
Poster published on 28 May 1992
cover (1992)
poster (1992)
In 1993 the Nicotra research group published the article about azasugars (also known as iminosugars):
Lay L., Nicotra F., Paganini A., Pangrazio C., Panza L.
A new procedure for the synthesis of azasugars
(1993) Tetrahedron Letters, 34 (28), pp. 4555-4558.
A new procedure for the synthesis of azasugars
Documents that cite the paper
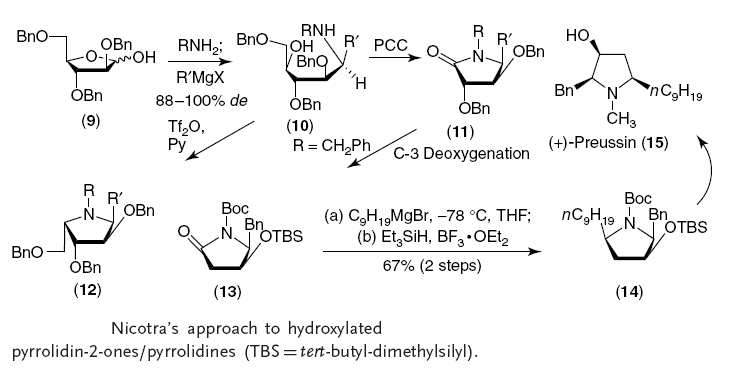
These authors have shown, for the first time, that the addition of Grignard reagents to N-benzyl and N-alkyl glycosylamines derived from perbenzylated pentofuranoses or hexopyranoses followed by a simple cyclization procedure afforded a short and convenient approach to imino-C-glycosides in the pyrrolidine and piperidine series. The procedure is illustrated in Scheme 2 (see compounds 16) from a d-arabinofuranosylamine, using octylmagnesium bromide, and from a d-glucopyranosylamine using allylmagnesium bromide, and cyclization promoted by reacting the intermediate amino alditol with triflic anhydride.
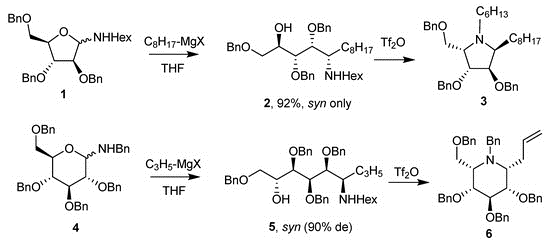

A first application of this reaction to the synthesis of azasugars was described in 1993 by the same authors, which opened the route to a new and efficient synthetic procedure towards this type of compounds.
This observation opened the way to an easy formation of azasugars following the sequence of a) glycosylamine formation, b) Grignard reaction and c) cyclization with Tf2O. This procedure is easily extended to include pyranose rings.
The synthesis of iminosugars via additions to glycosylamines was pioneered by the work of Nicotra and co-workers in the early 1990s. Several research groups have adopted Nicotras method for the synthesis of a wide variety of iminosugars with different configurations and substituents
The addition of organometallic compounds to glycosylamines introduced by Nicotra et al. is an efficient and versatile method to obtain functionalized pyrrolidines. The synthetic applications of this reaction have been demonstrated both by our group and by others for the synthesis of glycosidase or glycosyltransferase inhibitors.
Many synthetic approaches have been described for the preparation of iminosugars, including chemical and enzymatic methods. Among these, the procedure initially described by Nicotra involving amination of the anomeric carbon followed by treatment with a Grignard reagent and subsequent cyclisation is an easy process which has widely been used for the preparation of functionalised pyrrolidines.
In 2003, Yoda and coworkers reported an asymmetric synthesis of penaresidin B from D-arabinose derivative.Their synthesis relies on a route developed in 1993 by Nicotra et al. for the synthesis of azasugars following an oxidative degradation pathway.
For a review of the procedure, see:
The Chemical Synthesis of the 1-C-Alkyl Substituted Pyrrolidine and Piperidine Iminosugar Natural Products and their Analogues
New asymmetric strategy for the total synthesis of naturally occurring (+)-alexine and (-)-7-epi-alexine
Synthesis and Biological Activity of Penaresidins A and B, Penazetidine A, and Related Analogs
Glycoside Mimics from Glycosylamines: Recent Progress
Inhibition of Chitin Synthetase from Saccharomyces cerevisiae by a New UDP-GlcNAc Analogue
Synthesis of Glyphaeaside C and Structural Revisions of the Glyphaeaside Alkaloids
Wikipedia Iminosugar
Development of iminosugar-based glycosidase inhibitors as drug candidates for SARS-CoV-2 virus via molecular modelling and in vitro studies
|
|
|